These presentations provide short discussion of current topics and may be helpful to you in your practice. 
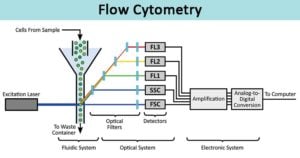
Jevremovic describes the use of flow cytometric analysis of bone marrow for the presence of dysplasia in chronic myeloid neoplasms.Welcome to Mayo Medical Laboratories Hot Topics. Dragan Jevremovic, Co-Director of the Cell Kinetics Laboratory at Mayo Clinic in Rochester MN. To learn more about analyzing T cells and other cell types by flow cytometry, and to get access to all of our advanced materials including 20 training videos, presentations, workbooks, and private group membership, get on the Flow Cytometry Mastery Class wait list.Our speaker for this program is Dr. However, advances are being made daily and eventually labeling single Treg cells correctly will be possible. The key is to test these populations’ functionally after identifying them because currently it is difficult if not impossible to define a single cell as a Treg. Using flow cytometry to first gate on and sort viable cells with markers consistent with Tregs, then functionally testing to see if, as a group, the cells defined by your gating strategy actually act like Tregs, is currently the best way to quantify Tregs in your sample.īy executing the right gating strategies for defining Tregs by flow cytometry and accounting for the growing number of Treg subsets, you can tease out your Treg populations of interest. Thinking again of the potential ‘effector T cell’ wolves in sheep’s clothing, we just don’t know how many non-suppressive, even inflammatory cells are hiding in our Treg gating strategy. Currently the only way to test Treg suppressive function is in a bulk culture, where one can conclude that some (but not all, possibly not even most) of the cells designated as Tregs are suppressive. However, to show a single cell is a Treg we ideally want to be able to quantify that this one chosen cell can suppress the function of effector T cells (or other cell subsets) in co-culture. Defining A Single Cell As A Treg-Is It Possible?Ī major limitation in the Treg field is the lack of a single cell suppression assay.ĭefining an individual T cell as a member of a distinct memory lineage, such as Th1, Th2, or Th17, can be accomplished via analyte analysis with single cell resolution such as intracellular cytokine staining, as these cells are primarily if not exclusively defined by what cytokines they produce. Also, in both mice and humans, Helios has been found to reliably distinguish tTregs from the p, and iTreg subsets. For instance, in humans, CD39 is considered a reliable tTreg marker. There are makers associated with these various Treg subsets and they should be considered for inclusion in a Treg anti body panel if subsetting them is of interest. Another flavor, known as pTregs, are generated in the periphery, and iTregs are induced in culture via TGF-b. There are many flavors of Tregs, including tTregs, pTregs, and iTregs.įor example, tTregs (also known as nTregs) are generated in the thymus and have a TcR repertoire that is biased towards self-peptides. Typically FOXP3 cannot be included in panels where viable cells are required post-sort as intracellular staining is required.ĭefining The Increasing Variety Of Treg Subsets In order to determine if the cells you are defining as Tregs functionally resemble them, Option 2 (see below) includes omitting FOXP3 from your panel, sorting CD3+, CD4+, CD25hi, CD127lo cells, then determining the functions of your ‘Treg’ population via cytokine analysis and/or suppression co-culture assays with non-Treg T cells (CD3+ CD4+ CD25-, CD127hi). Therefore, when relying on flow cytometry phenotyping alone to define Tregs, inflammatory T cells could be a wolf in sheep’s (Treg’s) clothing and lead to incorrect data interpretation.Ī cell may look like a duck, but does it quack? Measurement of the effector functions of your possible Treg population will greatly help elucidate the accuracy of your flow gating strategy. However, activated T cells often up-regulate CD25, and FOXP3 expression has been found on ‘effector’ (non-suppressive) T cell lineages. Using these markers, a clear population is often visible from samples such as mouse splenocytes and human PBMC. When looking solely at antigen expression, Tregs are often defined as CD3+, CD4+, CD25hi, FOXP3+, and CD127lo (shown in the figure below as Option 1). The standard Treg gating strategy for both mouse and human samples (after first gating out doublets and gating on live cells) includes the antigens CD3, CD4, CD25, FOXP3, and CD127. Written by Jennifer Snyder-Cappione, Ph.D. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
